 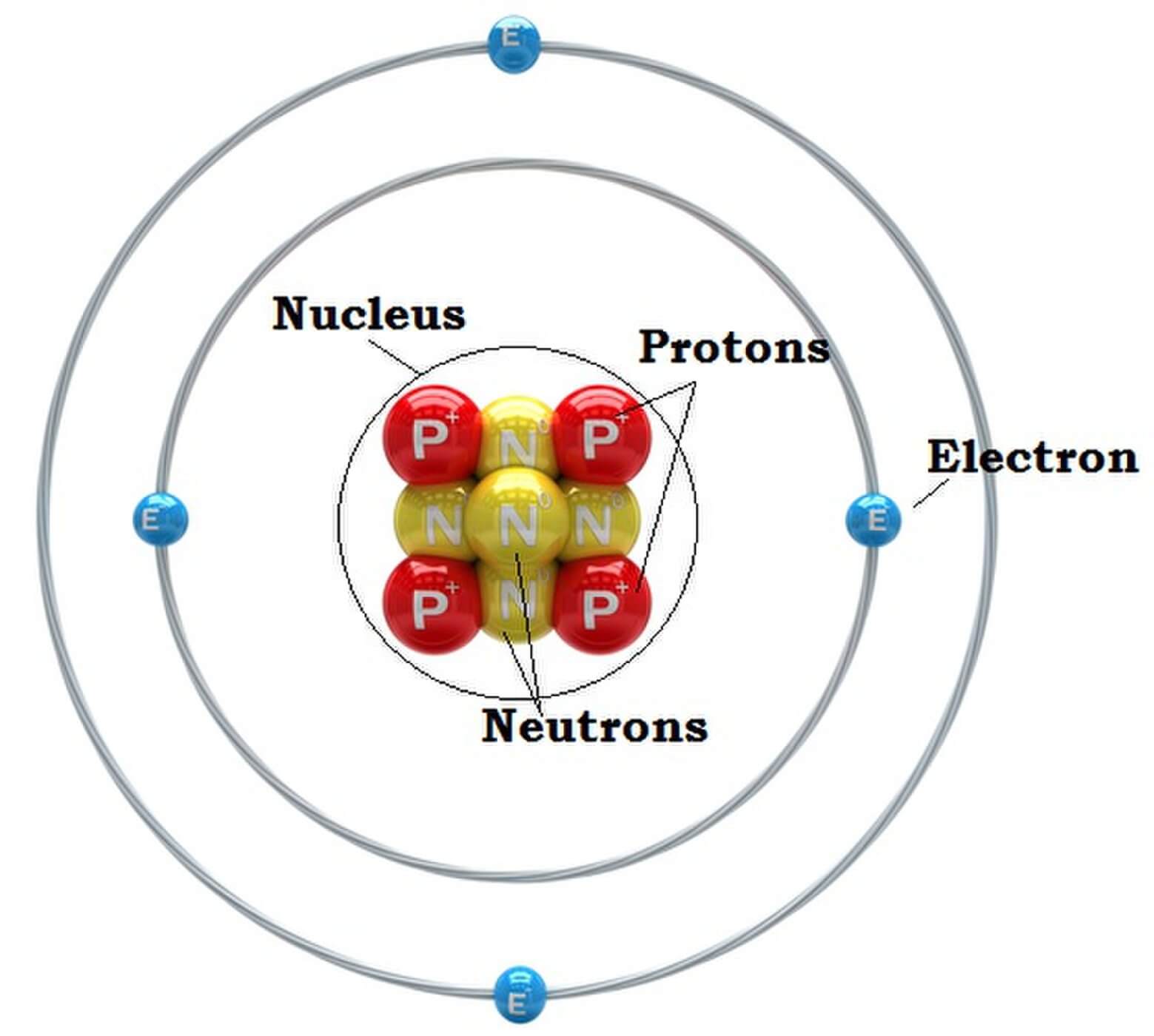
That's why the neutrons in the table below are labeled \(n0. Unlike protons and electrons, which are electrically charged, neutrons have no charge they are electrically neutral. Atoms of all elements (except for most atoms of hydrogen) have neutrons in their nucleus. 
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. Neutrons are composite particles made up of three smaller, elementary particles called quarks, held together by the Strong Force. The last of the three main particles that make up the atom is the neutron. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. You can use the links below to jump into the Help Documentation for Neutron 3: Introduction. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. Condition: New Model: Neutron 3 Elements Country/Region of Manufacture: United States Type: DAW/Multitrack Distribution Media: Hardware Key.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
